50 years of single channel recordings: one pore at a time, through time
In this 50th anniversary year of the foundational paper by Neher and Sakmann, our latest Current Affairs discusses the origins and importance of single channel recordings, defining key developments and texts through those five decades of single channel discovery, and offers a glimpse of future higher throughput potential.
Origins of the patch-clamp revolution
Imagine the ability to ‘see’ the activity of a single ion channel: its opening, closing, response to the body’s signalling molecules and drugs at millisecond timescales. Single channel recording is that ability: it gives the highest-resolution, real-time view we have of ion channel function, allowing researchers to observe the opening and closing of individual ion channels or pores.
Single channel recordings stem from the patch-clamp technique developed by Erwin Neher and Bert Sakmann (1976), who were awarded the Nobel Prize in Physiology or Medicine (1991) for enabling direct measurement of ionic currents through single channels. Their work transformed electrophysiology by making it possible to link single channel molecular events to electrical behaviour at the membrane level.
Hamill et al.’s classic methods paper (1981) remains the seminal work (and likely most cited electrophysiology paper ever) for achieving low-noise, high-resolution recordings.
Single-channel recording revelations
Unlike their robust, boisterous, bigger-brother whole-cell currents, the dainty, delicate, and detailed single channel recordings resolve unitary current amplitudes and the stochastic transitions between open and closed states. From these current traces, researchers can determine single channel conductance, ion selectivity, voltage or ligand dependence, and intricate gating kinetics such as burst behaviour or ‘flicker block’. Such parameters allow the construction of kinetic models that describe how channels move between conformational states, providing a direct structure-to-function translation.
For rigorous theory and analysis, the definitive, undisputed heavy-weight reference is the Single-Channel Recording Book, edited by Neher and Sakmann (1995). Complementing this, the statistical frameworks developed by Colquhoun and Sigworth (1995) remain essential for single analysis and kinetic modelling.
Perhaps, for a more accessible perspective on why kinetic analysis remains central to modern pharmacology, we point the reader to Sivilotti and Colquhoun (2016).
Strengths and limitations of the technique
The principal strength of single-channel recording is mechanistic resolution at sub-millisecond timescales. For instance, it allows investigators to distinguish whether a compound reduces current by blocking the pore, altering conductance, stabilising closed states or decreasing open probability. This level of detail is unmatched by whole-cell current (ensemble) recordings.
The trade-offs are practical. Single channel experiments are technically highly demanding, require stable gigaseals and very low-noise conditions; consequently, they are inherently very low throughput. Data acquisition and analysis are time-intensive and expertise essential for reliable interpretation.
For a broader overview of patch-clamp configurations, applications and limitations, see the review by Zhao et al. (2009).
Mechanistic insight in drug discovery
Despite the challenges, single channel recording is increasingly valuable in drug discovery because it reveals mechanism of action rather than simple drug activity and efficacy. Changes in open probability, burst duration or unitary conductance can aid explanation of potency, state dependence and therapeutic specificity.
A recent example is the work by Rodrat, Charlick and Sheppard (2026), which used excised single channel recordings to show how CFTR modulating drugs stabilise the F508del mutant chloride channel and enhance gating. These experiments provided a direct mechanistic basis for the clinical benefit of Vertex’s cystic fibrosis combination therapies (e.g. elexacaftor-tezacaftor-ivacaftor, Trikafta) by distinguishing effects on channel stability from effects on gating kinetics. This type of mechanistic clarity is particularly important for complex targets where multiple functional defects coexist.
From detail to scale: integrating patch clamp automation
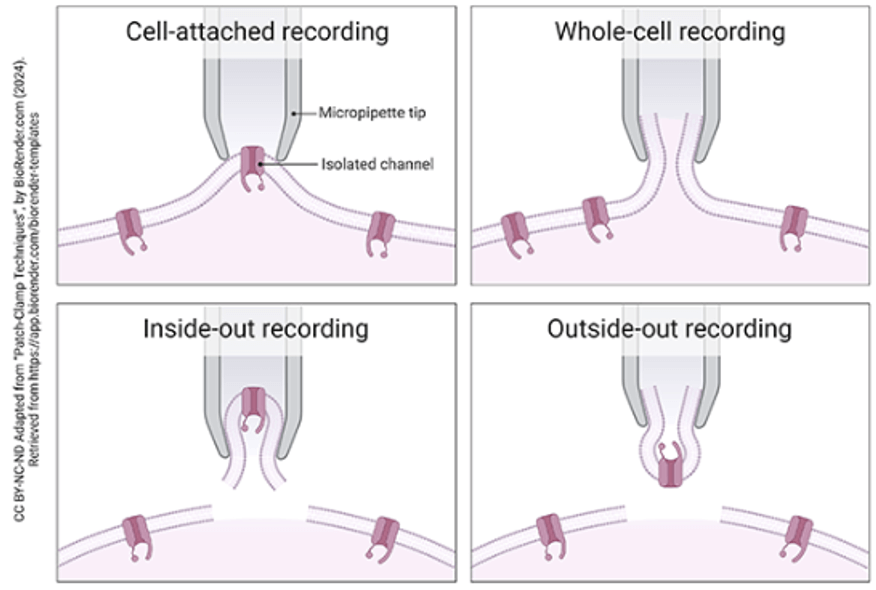
Automated patch clamp (APC) has transformed ion channel screening by enabling population-level pharmacology at far higher throughput. However, APC typically enables whole-cell measurements and does not always capture the temporal resolution or patch configurations (see Figure 1) required for detailed single channel analysis.
Figure 1 – Patch clamp recording configurations
An emerging strategy is integration and complementarity rather than replacement. APC can rapidly profile large compound libraries and identify functional hits, after which targeted single-channel experiments define mechanism, state dependence, and kinetic effects. Developments at Sophion will allow the existing high-throughput screening of whole-cell current functional hits, complemented by the single-channel mechanistic studies, both via APC. Thus, we are aiming to scale the previously slow, labourious, and technically challenging traditional single channel recording technique.
Figure 2. BK single-channel recordings made on QPatch APC
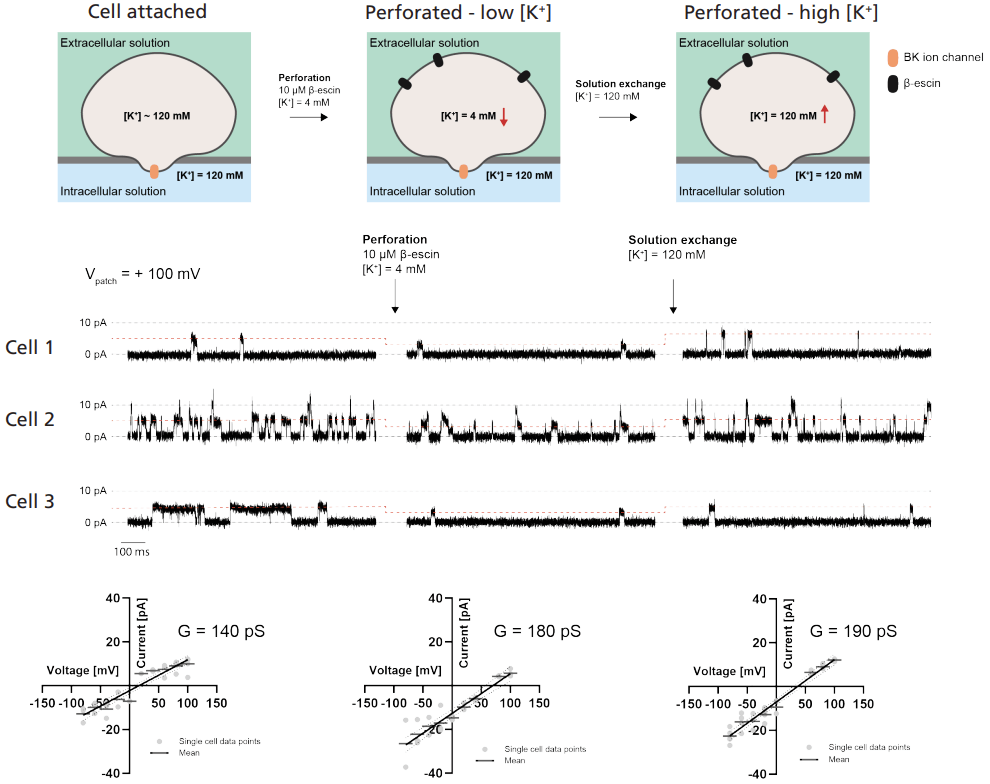
Copied from the Sophion poster “Advancing Automated Electrophysiology: Single-Channel Recordings in the Cell-Attached Configuration”.
Why single channels still matter and future potential on APC
As structural biology and computational modelling continue to advance apace, the need for functional validation at the level of individual ion channels is growing. Single channel recording provides the quantitative kinetic information required to test structure-function hypotheses and to understand how drugs reshape the energy landscape of channel gating.
In modern workflows, the greatest impact comes from combining scale with resolution: automated systems to explore chemical space efficiently, followed by single channel recordings and analyses to reveal how and why a compound works.
One pore at a time still delivers the clearest, cleanest answers, and when integrated with higher-throughput APC technologies, it remains one of the most powerful tools for mechanistic ion channel research and rational drug discovery and development.